Modulating biological aging with food-derived signals: a systems and precision nutrition perspective
Several factors, including diet, physical activity, and social relationships, influence aging and the development of NCDs. Research on centenarian populations suggests that plant-rich diets, balanced energy intake, and regular physical activity are linked to lower risks of chronic diseases and improved quality of life in older age11. These findings are further supported by recent evidence from two major prospective US cohorts (Nurses’ Health Study and Health Professionals Follow-up Study) involving over 100,000 participants followed for 30 years. These findings extend existing knowledge by linking diet adherence to multi-domain aging metrics, including cognitive and metabolic function. The studies showed that higher adherence to several dietary patterns, including AHEI (Alternative Healthy Eating Index), aMED (Alternative Mediterranean Index), DASH (Dietary Approaches to Stop Hypertension), and PHDI (Planetary Health Diet Index), was strongly associated with increased odds of achieving healthy aging12. This included preserved cognitive, physical, and mental function, as well as freedom from chronic diseases into older age. Among these, the AHEI showed the strongest association, nearly doubling the odds of healthy aging compared to the lowest adherence quintile.
While structured dietary patterns such as the Mediterranean diet or AHEI demonstrate robust associations with healthy aging, their effects emerge from the complex interplay of numerous food-derived bioactive compounds. Precision targeting of specific molecular pathways, for instance, those modulated by polyphenols, omega-3 fatty acids, or vitamin D3, can enhance our mechanistic understanding and intervention strategies. Rather than an either/or dichotomy, whole-diet frameworks and molecularly targeted approaches should be viewed as synergistic components of a unified strategy for healthy aging.
Beyond lifestyle factors, emerging research highlights the role of biological markers in assessing aging trajectories. One of the most promising tools in this area is the use of epigenetic clocks. Epigenetic factors, including immunomodulating micronutrients like vitamin D3, play a significant role in aging13. They support immune competence, reduce chronic inflammation, optimize metabolic pathways including management of oxidative stress, and help maintain a balanced gut microbiome, all of which contribute to overall health and longevity. Recognizing the interplay between lifestyle, social environment, and genetic predisposition is crucial for developing comprehensive strategies to support healthy aging at both individual and population levels14.
Most epigenetic clocks rely on bulk tissue samples such as blood, which contain varying cell types that shift with age. This heterogeneity can obscure true aging signals. It is thus important to distinguish between extrinsic clocks (influenced by cell composition) and intrinsic clocks (corrected for cell-type proportions). A clear distinction between chronological and biological age is essential for advancing aging research15. Epigenetic clocks, which estimate biological age based on DNA methylation patterns, can be broadly categorized into several types (Table 1). These are chronological clocks (e.g., Horvath16), which align closely with actual age; biological risk clocks (e.g., GrimAge17), which predict health outcomes and mortality risk; mitotic clocks (e.g., epiTOC218), which track cellular replication; and noise barometer clocks, which capture stochastic variation in methylation patterns. Each category serves distinct purposes, from assessing disease risk to evaluating the impact of interventions, making the choice of clock crucial for accurately interpreting aging trajectories17,19. Moreover, recent research indicates that part of the variance in epigenetic clock rates may be genetically determined20.
The selection of a clock should align with the research objective. For instance, GrimAge is particularly well-suited for evaluating NCD risk and mortality prediction17. Since the introduction of the first epigenetic clock, the field has rapidly expanded, with numerous clocks now developed using diverse algorithms and data modalities19. Beyond epigenetics, aging clocks have also been constructed using taxonomic profiling of the gut microbiome, offering additional insights into host aging. Moreover, circadian rhythms within the gut epithelium are influenced by feeding patterns and microbiota-derived signals, which interact with host immune and neural systems21. This suggests that maintaining rhythmicity in microbiome-host interactions may be an additional target for interventions aimed at delaying cognitive aging. Further complexity arises from recent findings showing that plasma proteomic signatures can define organ-specific clocks in living individuals, opening new avenues for precision aging diagnostics22.
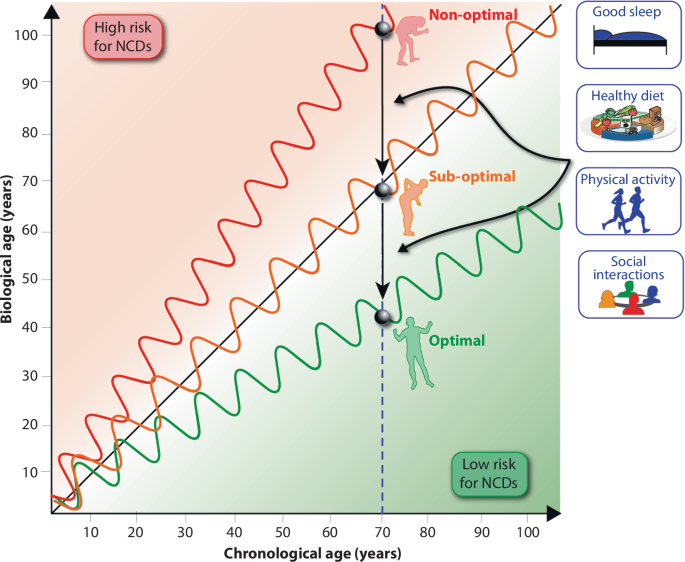
Figure 1 illustrates the concept of biological age and how it differs from chronological age in individuals. Chronological age refers to the actual age in years, while biological age reflects how well an individual’s body functions compared to others of the same chronological age. Factors such as genetics, lifestyle, and environmental influences contribute to this variation. In the figure, three hypothetical individuals are shown at the age of 70 (represented by circles). Despite having the same chronological age, their biological age varies. This is visually represented by three different trajectories:
-
Optimal aging (green line): This individual’s biological age remains below chronological age, reflecting good health, a balanced diet, regular physical activity, and minimal chronic diseases.
-
Sub-optimal aging (orange line): This individual’s experience moderate biological aging, possibly due to poor dietary habits or lack of physical activity, leading to early signs of aging and the onset of chronic health conditions.
-
Non-optimal aging (red line): This individual’s biological age exceeds their chronological age, indicating accelerated aging, likely influenced by unhealthy lifestyle choices such as smoking or other forms of intoxication, poor diet, physical inactivity, and increased risk of age-related diseases like cardiovascular diseases or diabetes.

Biological age, as opposed to chronological age, reflects the functional status and health risk of an individual. The diagonal dashed line represents chronological aging. Three illustrative aging trajectories, optimal (green), sub-optimal (orange), and non-optimal (red), demonstrate how biological age can diverge depending on lifestyle choices. Individuals aging optimally maintain a biological age below their chronological age and exhibit lower risk for NCDs, while those with non-optimal aging experience accelerated biological aging and higher NCD risk. Wave patterns represent short-term variability influenced by daily and seasonal factors. Arrows indicate how targeted interventions, such as good sleep, a healthy diet, regular physical activity, and social engagement, can decelerate aging and shift individuals toward a more favorable aging trajectory.
This emphasizes the role of lifestyle factors in influencing biological aging, with diet, exercise, sleep quality and social interactions serving as key factors in maintaining health and slowing down biological aging. This concept is critical to understanding how we can intervene to delay aging and prevent chronic diseases. Diet is a significant factor in influencing biological age. A balanced diet rich in nutrients, such as antioxidants, fiber, and polyunsaturated fats like omega-3 fatty acids, can help slow down the progression of aging. Conversely, poor dietary choices, such as high sugar, trans-fats, and low fiber, accelerate the aging process, increasing the risk of NCDs.
A plant-based diet containing high quality protein is associated with a 15% reduction in the risk of cardiovascular diseases and a 21% reduction in coronary artery disease23,24. Mechanistically, this is based on pathways like the inhibition of mTOR (mammalian target of rapamycin), modulation of sirtuins, and reduced oxidative stress. Moreover, adherence to the Mediterranean diet has been linked to reduced inflammation and improved cognitive function, with studies indicating a lower risk of cognitive decline and dementia among its adherents25,26. Vegan diets have been shown to improve glycemia and weight management for individuals with type 2 diabetes24. However, plant-based diets may lack essential micronutrients like vitamin B12, vitamin D3, iron, calcium, and long-chain omega-3 fatty acids. Ensuring adequate intake through careful dietary planning or supplementation is essential.
Furthermore, maintaining a healthy weight through a balanced diet and regular exercise can prevent or delay many chronic conditions, such as hypertension, diabetes, and heart disease. Interestingly, also social engagement and strong familial ties, as seen in Blue Zone populations, contribute significantly to longevity and overall well-being27. In contrast, loneliness has been associated with a poorer diet quality increasing the risk of malnutrition.
link






